Anodised Aluminium Processing Observation and Measurement Using Digital Microscopes
A thin oxide film (approximately 2 nm) naturally forms on aluminium when it comes into contact with air. Therefore, it is commonly believed that aluminium does not rust easily. However, this film is extremely thin, so chemical reactions may cause aluminium to corrode depending on the environment. Anodised aluminium processing is a type of surface processing in which this oxide film is artificially formed on aluminium. This section provides an overview of anodised aluminium processing and introduces observation and measurement examples using a digital microscope.

- What Is Anodised Aluminium Processing?
- Anodised Aluminium Formation Process and Structure
- Coloured Anodised Aluminium and Hard Anodised Aluminium
- Advantages and Disadvantages of Anodised Aluminium Processing
- Anodised Aluminium Processing Observation and Measurement Examples Using a Digital Microscope
What Is Anodised Aluminium Processing?
Anodised aluminium processing is a type of surface processing in which an anodic oxide film is formed on the surface of aluminium.
Plating is another type of surface processing, but whereas plating causes a film of another metal to form on the aluminium surface, anodised aluminium processing causes a growth film to form on top of and a penetrating film to form underneath the aluminium surface.
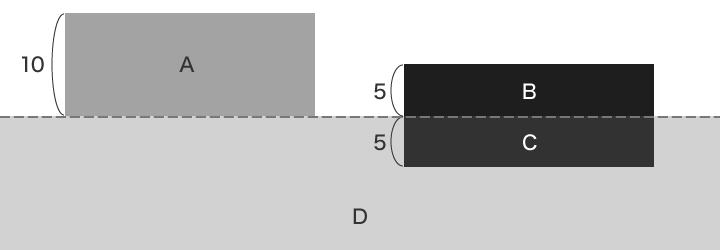
- A: Plating layer
- B: Growth film
- C: Penetrating film
- D: Aluminium material
Half the anodised aluminium film grows on top of the aluminium surface. The other half penetrates into the aluminium surface.
Anodised Aluminium Formation Process and Structure
Passing an electric current through aluminium causes the microscopic dents and protrusions on the surface to dissolve (penetrating into the surface) while at the same time causing an oxide film to form. As time passes, a 3D structure known as a cell forms.
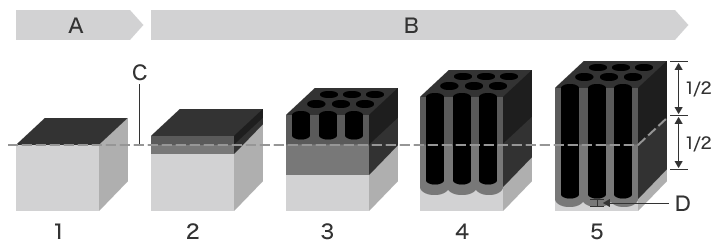
- A: In air
- B: In electrolytic solution
- C: Original aluminium surface
- D: Barrier layer
- In air, an oxide film approximately 2 nm thick forms naturally.
- The barrier film grows.
- Holes 10 to 20 nm thick open up.
- Oxidation and the dissolution of the film proceed simultaneously, elongating the holes.
- The film grows proportionally to the time electrolysis is applied.
Coloured Anodised Aluminium and Hard Anodised Aluminium
Normal anodised aluminium can be coloured by sealing organic paint in the microscopic holes formed on its surface. Coloured anodised aluminium is not commonly used in building materials because while it is resistant to easy removal of the colouring, it is vulnerable to ultraviolet rays and heat.
With a thick oxide film, hard anodised aluminium is harder than normal anodised aluminium. Hard anodised aluminium is used in sliding parts (shafts and rolls), automotive engine parts, aircraft parts, and similar applications. This is because it has excellent hardness, wear resistance, corrosion resistance, insulation properties, and heat resistance.
| Comparison item | Normal anodised aluminium | Hard anodised aluminium |
|---|---|---|
| Colour | White or coloured | Grey (generally cannot be coloured) |
| Hardness | Approximately 200HV | 400HV or more |
| Film thickness | 5 to 25 µm | 20 to 70 µm |
| Applications | Building materials, household goods, decorations | Sliding parts (shafts and rolls), automotive engine parts, aircraft parts |
Advantages and Disadvantages of Anodised Aluminium Processing
This section explains the advantages and disadvantages of anodised aluminium processing.
- Advantages
-
- Corrosion resistance: The corrosion resistance is improved.
- Insulation properties: The insulation properties are excellent because the oxide film does not conduct electricity.
- Thermal conductivity: The thermal conductivity is a third that of aluminium that has not undergone anodised aluminium processing.
- Hardness: Although normal aluminium has a hardness of 20HV to 150HV, aluminium that has undergone anodised aluminium processing obtains a hardness of 200HV or higher.
- Colouring: Various colours can be applied to the microscopic holes on the surface.
- Disadvantages
-
- Heat resistance: The surface will crack or peel in environments whose temperature exceeds 100°C.
- Fragility: The surface is not flexible, so it will crack or peel if bent.
Anodised Aluminium Processing Observation and Measurement Examples Using a Digital Microscope
These are the latest examples of anodised aluminium processing observation and measurement using KEYENCE’s VHX Series 4K Digital Microscope.

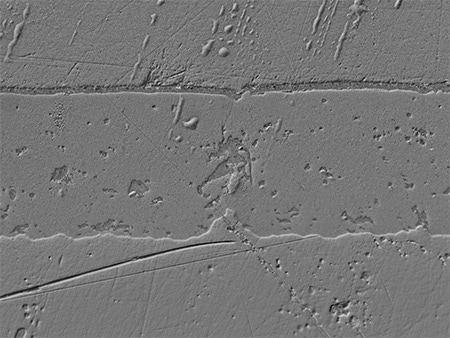

Optical Shadow Effect Mode provides a clear picture of the surface texture.

Conventional SEMs were utilised for this measurement; however, sample preparation required a significant amount of time.


The glare removal function allows for
observation without glare.
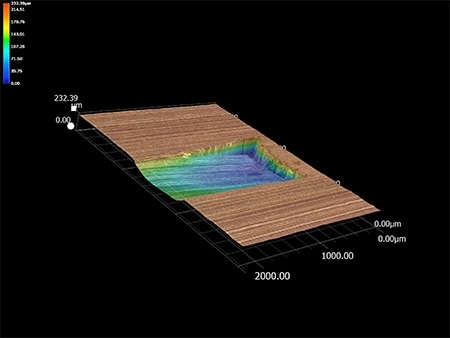
The 3D function enables defect cause identification by showing how scratches are formed.